|
|
 |
 |
|
| |
Hologram
formation control in the thick dichromated
gelatin-glycerol
systems
Alexander Malova, Nadya Reinhand*b, Yury Vigovskyc, Yulia Zagainovaa
aIrkutsk State University, Gagarin bul., 20, Irkutsk, 664003, Russia
bInstitute of Mechanical Engineering Problems of the Russian Academy of Sciences,
V.O., Bolshoi pr., 61, St.Petersburg, 199178, Russia
cMeDia Ltd., Novoslobodskaja str. No 31/1, Moscow, 103055, Russia
ABSTRACT
Selfdeveloping dichromated
gelatin (SD DG) is holographic photosensitive medium that
possesses the number of unique properties. Being illuminated
by interferometric pattern it records the energy distribution,
and in such way the gratings with high diffraction efficiency
can be obtained. The absence of postexposure processing allows
to use it for in-situ experiments. And besides the selfdeveloping
property of the material allows to create new kinds of optical
elements based on very thick gratings that possess very high
selectivity properties.
The mechanism of the hologram
formation in the material is proposed and the possibilities
to control this process are considered. The recorded hologram
is the result of conformational changes in the structure of
a gelatin system. It is the result of hierarchy
of sequential structural gelatin macromolecules modifications.
Characteristics of these processes can be effectively controlled
at the levels of the primary (the chemical composition of
emulsion) and ternary (coil-globule transitions for the entire
macromolecule) structures of the SD DG system. We analyzed
the influence of IR
laser annealing and/or special highly hardened gelatin sublayer
on the gelatination acceleration and resulting diffraction
efficiency. of layers with thickness more than 1 mm. Salt-doped
SD DG layers were experimentally tested. The
properties of new version of SD DG emulsion containing organic
high-molecular-weight polymer are discussed.
Keywords: dichromated
gelatin, glycerol, selfdeveloping, volume holograms, red sensitivity,
collapse.
- INTRODUCTION
The holograms registered in photosensitive layers of several millimeters thickness have a number of specific properties, the most important of which are extremely high spectral and spatial selectivities1. These highly selective gratings are used for the development of modern optical information processing devices, in optical memory systems with multiplex information recording and for interfiber connectors in optical telecommunication networks, for the variety of spectral filters for imaging, etc.2,3.
Usual holographic materials can not be applied for recording of grating of millimeter thickness, because it is impossible to maintain the material uniformity during the post-exposure processing, Differential shrinkage caused by post-processing leads to the reduction of diffraction efficiency, and besides to the asymmetry and widening of selectivity contour of output signal1.
Selfdeveloping dichromated gelatin4 is one of the solutions to overcome these problems because it does not require post-exposure processing. The developed photosensitive medium differs from the wellknown dichromated gelatin by the addition of certain amount of glycerol. Glycerol in these layers serves as the developing component and also as the plastic component. It keeps some amount of water molecules that are carrying out the developing process due to their hydrogen ties. And besides glycerol provides enough plasticity to the material to make layers of millimeter thickness.
However several problems arise at synthesis of superthick (more than 0,5 mm) layers. We have to find the reasonable limit of the emulsion drying time. Very long drying (more than three day) is preferable, it leads to better gelatination and "ripening" of colloidal medium. However, long term drying is not suitable from the technological point of view. Our attempts to solve this by conventional methods such as ventilation by hot air or alcohol dehydration were not successful, they result in originating of a strong gradient of optical refraction in the layer depth. The processing of colloidal layers by microwave radiation resulted in their flaking from the substrate and to unmonitored change of the conformation gelatin macromolecules status.
- GELATIN STRUCTURE AND HOLOGRAM RECORDING
It is wellknown that highly efficient holograms are recorded by means of creation of refractive index modulation inside the materials. Usually it is achieved by specially organized phase transition in a substance. There are two main requirements to any recording media: high photosensitivity and high spatial resolution. One can control the material photosensitivity by preparing the recording media where phase transitions are characterized by an appropriate inversion degree of molecules (in the first-order approximation, such molecules can be considered as two-level quantum systems). Simultaneously, a threshold character of the relevant photochemical reaction should be ensured which is necessary for the pre-exposure storage of the photomaterial. To achieve a high spatial resolution of the recording medium, the condition of spatial localization of the primary photochemical reaction should be met. If the requirements specified above are met, optical radiation being recorded plays the role of a "trigger" which actuates the mechanism of changes in the phase state of the recording medium at micro- and macrolevels.
In this paper, we invoke the biophysical data5,6 to consider the mechanism of the recording of optical data in SD DG layers as a set of hierarchical interrelated phase transitions at various levels of gelatin structure, including the intramolecular, molecular, and macromolecular levels.
- Cascade of phase transitions
The main distinguishing feature of dichromated gelatin as a polymer is associated with the natural origin of gelatin, which is produced as a result of the denaturation of collagen which macromolecules have a structure of triple hyperhelix. Therefore, the native state of gelatin macromolecule also has a biohelix-like structure, which makes it possible to prepare DG layers with an initially ordered (helical) molecular structure. In the process of development, the phase transition from the ordered (quasi-crystalline) state to disordered (quasi-amorphous) state occurs under the action of light and water at all the levels of structure organization of gelatin layer, which leads to large photoinduced changes in optical characteristics of a DG layer.
In other words, the synthesis of dichromated gelatin layer before the recording process is accompanied by the appearance of stresses applied to "springs" representing the helical fragments of gelatin macromolecules. Then, the action of light plays the role of' a trigger, which sets the springs free. The energy stored in these springs induces a cascade of phase changes in the state of gelatin layer. These phase changes, in their turn, give rise to variations in the macroscopic characteristics of the recording medium. Note that, apparently, artificial polymers, for example, polyvinyl alcohol, do not exhibit such clearly pronounced native structure and therefore, cannot compete, at least in holography, with dichromated gelatin layers.
2.2. The structural organization of the dichromated gelatin systems
Gelatin is a highly asymmetric linear polypeptide polymer of protein nature. Macromolecule of gelatin consists, on the average, of 500-600 amino acid radicals. The molecular mass of gelatin macromolecules ranges from 40,000 to 1000,000. Similar to all the proteins, gelatin is a high-molecular compound characterized by its ability to form various supermolecular structures5.
There are several basic levels in the structure organization of proteins. The primary structure is determined by the composition and the sequence of amino acid radicals in a macromolecule of the polypeptide chain. The secondary structure is determined by the configuration and relative spatial arrangement of protein macromolecules, which have the shape of helixes, folds, or globules. The hypersecondary structure is determined by the aggregation of secondary -structure elements into a single macromolecule, which is manifested, in particular, as the phase state of the globular nucleus. The domain structure is determined by external parameters (the thickness of layer, the degree of adhesion of the layer to a substrate, etc.) responsible for the formation of separate and relatively weakly bonded globular segments of macromolecule. The ternary structure is determined by the conformation of protein helixes in the form of fibrils or globules. The quaternary structure is determined by die aggregation of protein fibrils and globules and similar to the domain structure, it is highly sensitive to external conditions (the conformation of a collagen-like triple hyperhelix is the limiting case of the quaternary structure for gelatin macromolecules).
2.3. The control of the dichromated gelatin structure
Macroscopically, in the preparation of a DG layer, gelatin as a polymer system passes through a sequence of different aggregate states. The initial state in this sequence is the dilute solution of gelatin in water, where macromolecules reside in the state of Gaussian coil or globule. In the process of DG layer preparation, when the solution is poured onto a substrate, the interaction of chain macromolecules gives rise to the formation of a gel. This gel may exhibit properties of a liquid crystal in the case of rigid-chain molecules or properties of a strong solution with equal volume portions of the solvent and polymer5. As the solvent (water) is evaporated out of the emulsion poured onto the substrate, macromolecules return to their native state, forming a collagen-like triple helical structure. The deformation degree of this structure is determined by the conditions of film formation. In the case of gelling, the forces acting from the side of the substrate and conditions of drying lead to the unfolding of macromolecules into linear structures with simultaneous twisting of segments of these structures into helixes. Such a renaturation in the course of gelling has a statistical and local character and occurs through the linking of segments, giving rise, in its turn, to a clearly pronounced short-range order in the gelatin film. The long-range order, which is characteristic of collagen, is not observed in this case5. Depending on the thickness of emulsion applied to a substrate, films obtained by molding through gelling on solid substrates exhibit planar orientation of their structure elements. Obviously, the state of structure elements in the film depends on the state of these elements in the emulsion solution. Specifically, gelatin in a film obtained from solution at a temperature higher than 35 °C has a conformation of a Gaussian coil4-6 and does not display any features of ordering and planar orientation of structure elements.
We analyzed the possibility to control the emulsion adhesion to the substrate. The experiments showed that it can be controlled with the use of a gelatin sublayer with a thickness of 0.5-1 micron with different hardness. Thus, in synthesizing and pouring of SD DG layers, one can control the structure of gelatin layer.
It is important to take into account the influence of substrate properties in variety of applications, particularly, for interfiber connectors, where the substrate role is played by bridged fibers.
2.4. The water structure and it’s control by means of the IR laser annealing
The SD DG system contains water in quantity sufficient for the hologram development. The water in such system is held with the help of glycerol and consequently the holographic properties depend on glycerol concentration in a system7. This system contains great many of water, and, therefore it is possible to consider it almost as aqueous solution, to which the quasicrystal liquid structure role is essential8. The collective character of the water molecule motion is confirmed by the molecular dynamics results9. Under the IR radiation action the water structure energy increases and weakens hydrogen bindings, the oscillation frequencies increase, the concentration of defects which are generating at molecules translations from the site of a crystal lattice in an adjacent potential well is augmented. The water molecules form a collective module10. The interaction of water with solutes is described thus through aquacomplex, the aggregation of the material molecule with several water molecules. The water molecules, which are included in the complex structure, interact with adjacent water molecules. And it is possible to consider aquacomplexes as elements of hydrogen bindings net in a water5,9.
It is possible to influence on SD DG structure in all volume by means of the laser IR radiation. IR electromagnetic radiation causes molecular rearrangement in the material. This effect consists in the loss of coordinate oscillations stability: the part of molecules does not return in the position with initial coordinates. It is impossible to estimate the optimal radiation wavelength for such annealing because of composite emulsion structure. Therefore, basing on the known data on absorption spectrum of water we chose 1 micron wavelength radiation. Besides it has another advantage that the IR near range radiation passes through the glass substrate and consequently in thicker layer there are no undesirable effects of an interference of IR radiation.
Thus the energy pumping should be by very selective in order to change only tertiary and quaternary gelatin structures, causing their “elongation”. Therefore it is expedient to use laser radiation with small quantum energy and which is not absorbed by gelatin and single water molecules. The IR electromagnetic field action on an aqueous solution of gelatin macromolecules results in disturbance of equilibrium system condition. After the irradiation discontinuance there is a dielectric relaxation. Every relaxation, including dielectric, is the process of the system returning to equilibrium or system transition to the new status. After action of IR radiation, the water molecules start to be rebuilt according to Le Chatelier-Braun principle. Being bound in a unified hydrogen binding net, the molecules can be rebuilt only with the help of preferable movements. It means, that the time of this rearrangement (Debye time) should be much more, than time of one transition. The molecular movement in a three-dimensional spatial net of hydrogen bindings descends more often, than gaps of its connections to adjacent molecules. Thus the frequency of absorption 1000 cm-1 (wavelength is equal to 1 micron) can be interpreted as the sum of two frequencies: one of which is 200 cm-1 , the frequency of personal rotary oscillations, and ~ 800 cm-1, vibration frequency of water molecules in aquacomplex or collective module.
3. Experimental technique
3.1. Synthesis of selfdeveloping dichromated gelatin layers.
The procedure of the sample preparation is the following11. 12: the gelatin is solved during 1hour in water (1 g of gelatin on 5 ml of distilled water) at temperature 50oC, then glycerol (0.8 ml in 5 ml of water) was added, and the solution was maintained at temperature 40oC within 2 hours. Then we added ammonium bichromat of necessary concentration (20 % of the weight of dry gelatin or 0.2 g in 5 ml of a water) in the obtained homogeneous solution. Then ammonia for achievement of pH=9.0 was added, then the stain methylene blue (MB) as solution of 10 mg of the stain in 100 ml of water was added. The prepared emulsion solution filled up the transparent pan with lateral walls of the necessary altitude (from 0.5 up to 3mm), and it was covered by special glass. Gelatination procedure was carried out at the temperature of 25oC within 24 hours. In outcome the layer of the given depth was received.
KCl doped emulsions were tested to investigate the possibility of chemical control of the SD DG structure. KCl was added during the emulsion preparation in the amount of several percent.
We used the gelatin sublayers to investigate the influence of adhesion from substrate. The substrate was covered with 1% gelatin water solution. After drying the sublayer of about 1 micron thickness was obtained. Then it was hardened in the Orwo-400 solution during the various time intervals.
3.2. Technique of the laser annealing
After holding the emulsion layer during a day the quasicrystal selfdeveloping dichromated gelatin structure was formed. Then the plate was subjected to IR laser radiation with 1 micron wavelength and with known values of power in the pulse. In order to eliminate the influence of all transition processes, the hologram was recorded 10-12 hours later.
3.3. The holographic characteristics measurement.
We illuminated samples with interference pattern constructed by two plane light waves from the HeNe laser. The spatial frequency of the recorded diffraction grating was hundreds lines/mm. The energy of two beams was 6mW. Diffraction efficiency was measured as the relation of intensity values of diffracted and input beams.
- EXPERIMENTAL Results
4.1. The IR laser annealing
The variable parameters for the annealing IR pulses are the pulse energy, duration, and shape. It should be mentioned that maximal diffraction efficiency obtained in SD DG layers is about 70%. However when we analyzed the effect of different factors on the diffraction efficiency we were working in the range of 5-10% efficiency in order to eliminate the influence of non-linear properties of medium.
The experimental results for IR laser annealing are shown in Fig. 1 for holographic grating of 600 lines/mm recorded in selfdeveloping dichromated gelatin layer of 1mm thickness by radiation of HeNe laser. We analyzed the dependence of diffraction efficiency on the amount of IR laser annealing pulses and single pulse energy (a) and the best result occurred for five annealing pulses. Figure 1 (B) shows the obtained diffraction efficiency of holograms subjected to the IR laser annealing by five pulses depending on pulse duration and single pulse energy. One can see from this figure that there is an area of considerable diffraction efficiency increasing. The best result received at an annealing by five pulses with duration 2,5msec at impulse energy equal to 8-10 J. However, the exact energy level depends on the material thickness, on the concentration of gelatin in emulsion, and on the type and series of gelatin. It should be pointed out that all obtained values of diffraction efficiency for the annealed materials are higher (on average 2-3 times) than they are for the plates not subjected to the annealing. It is visible that the diffraction efficiency curve (Figure 1) has the brightly expressed peak (for annealing energy equal to 1,6 J) at small pulse duration. Further increase of pulse energy leads to considerable diffraction efficiency increasing. The diffraction efficiency increase in 5-6 times was observed for the pulse energy of 8J.
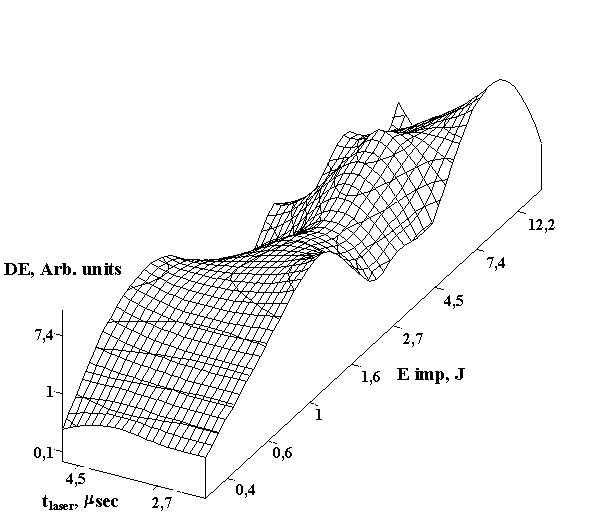
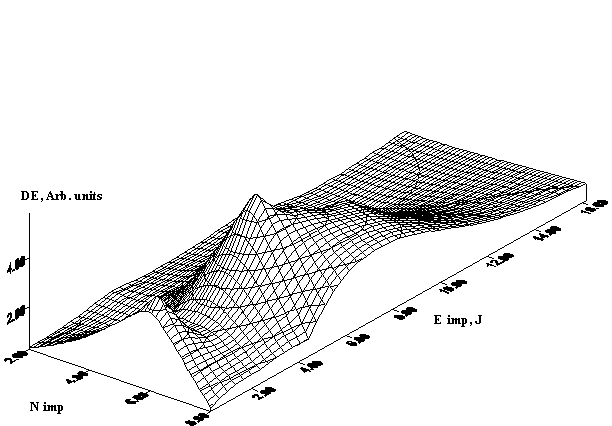
Figure. 1. Diffraction efficiency of holograms recorded in selfdeveloping dichromated gelatin layers with different energy and amount of IR laser annealing pulses (a) and with different energy and duration of five annealing pulses (b).
The pulse irradiation regime for laser annealing is preferable, because it allows to estimate precise energy of effect on the layer structure. The pulse irradiation is to some extent like sharp system “shaking”, and the necessary macromolecules packing is reached by the following relaxation processes in the system.
In our opinion IR electromagnetic field causes impulsive disturbance of water at the level of collective modules while single water molecules and gelatin squirrels do not absorb radiation of 1 micron wavelength9. Water quasicrystal net rearrangement conducts the change of electromagnetic field affecting gelatin macromolecules. These macromolecules move under of this field action and change therefore tertiary and quaternary conformation status to achieve an energy minimum in new quasicrystal net. Thus spiral segments do not change, and this means that the SD DG system photosensitive properties do not change too. (Our conception consists in the material photosensitivity based on untwisting of spiral segments.)
In general our experiments showed that the variety of pulse parameters affect on the resulting diffraction efficiency value. The main parameter is, of course, the amount of energy absorbed by the material. However, the pulse duration and its shape (this means the sharpness of fronts) as well as the moment of the annealing relatively the emulsion maturing time are also of importance. It is obvious from the experiments that there exist several solutions to reach the maximal diffraction efficiency by combining the optimal pulse energy, shape, duration and amount of pulses. However that concerns the moment of annealing, multiple experiments showed that the optimum is around 24 hours after emulsion pouring on substrate
4.2. The influence of adhesion to substrate on the selfdeveloping dichromated gelatin structure
Experiments showed that SD DG layer separation from glass substrate often occurs causing the diffraction efficiency reduction. This effect is called photoinduced collapse of the emulsion at recording. For elimination of this phenomenon we proposed to utilize an additional gelatin sublayer with the hardness higher than it is for the photosensitive emulsion layer. We investigated what optimal value of the sublayer hardness. The dependence of holographic properties of the SD DG layer on the substrate hardness is shown in Fig. 2. One can see from the Figure 2 that the maximal diffraction efficiency was observed for the sublayer subjected to hardening in ORWO-400 during 4.5 munites (corresponds to H=4.5).
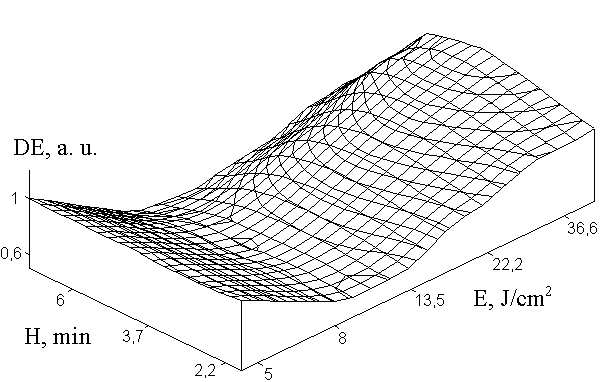
Fig. 2. Influence of hardening degree Н of sublayer on the SD DG exposition characteristics.
4.3. SD DG emulsions containing high-molecular-weight polymer
We developed the new version of selfdeveloping dichromated gelatin, in which 50 % of glycerol in emulsion was replaced by natural polymer obtained from plants, its molecular mass is about 20,000. The experiments showed considerable reduction of the layer gelatination time (Figure3). The formation of quasi-crystal structure occurs faster in this case compared with initial version of SD DG. Obtained holograms have improved storage time.
Fig. 3. Influence of the gelatination time on the SD DG exposition characteristics for 50% polymer replacement of glycerol.
4.4. The SD DG emulsion doped by KCl
We investigated the influence of salt addition to the composition. We used KCl, because it is known that K ions affect the aquacomplex structure. The experimental results are shown in Figures 4, (a) and (b). It is found that KCl essentially improves photosensitivity of the emulsion. The shape of the diffraction efficiency contours changes under the influence of KCl, however the character of this change depends on gelatination temperature schedule.
(a)
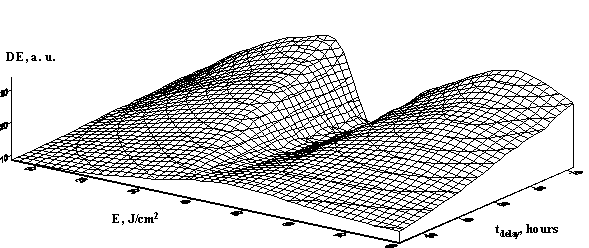 (b) (b)
Fig. 4. The hologram diffraction efficiency for SD DG doped by KCl in the gelatination time dependence at temperature 24о С (a) and at temperature 00 С (b).
5. CONCLUSIONS
We proposed different technologies to improve the holographic characteristics (diffraction efficiency, storage time) of selfdeveloping dichromated gelatin material.
It was shown that IR laser annealing (series of pulses) before hologram exposure results in increase of diffraction efficiency.
In order to overcome the problem of the photosensitive layer separation from the substrate we proposed to use strongly hardened gelatin sublayers. It was shown experimentally the improvement of diffraction efficiency in this case.
It was shown that the storage time of hologram can be improved by replacement of part of glycerol by natural polymer with high molecular weight.
Addition of salt to the emulsion leads to the increase of the material photosensitivity and to the diffraction efficiency improvement due the influence of ions on water molecules inside the material.
It should be pointed out that it would be reasonable to carry out further experiments with the usage of two lasers, blue and red ones, simultaneously, in order to substrate the influence of sensitizer.
6. ACKNOWLEDGMENTS
Our thanks for help in the experimental work to A.L.Antipov, I.V.Bogdan, L.E.Kruchinin, S.N.Malov, S.A.Medvedeva, V.Yu.Molotcilo, A.A.Petrov, S.N. Pidgurskii.
7. REFERENCES
- N.Reinhand, Yu.Korzinin, I.Semenova. “Very thick holograms: manufacturing and applications”, Journal of Imaging Science & Technology, 41, 3, 241, 1997.
- Yu.N. Kulchin. The distributed fiber-optical sensors and measuring nets., Dal'nauka Vladivostok 1999. ( in Russian).
- N.Reinhand, Yu.N.Denisyuk, N.N.Ganzherli, I.Maurer, I Pisarevskaya, V.Markov, “Application of selfdeveloping dichromated gelatin for holographic data storage”, Proceedings SPIE(Photonics West’98), 3294, pp.22-30, 1998.
- N.F.Balan, A.I.Erko, V.V.Kalinkin, A.N. Malov. et al, USSR Patent no. 1347757. 1985.
- .A.Yu. Grosberg, A.R. Khokhlov. Statistical Physics of Macromolecules Nauka, Moscow, 1989. (in Russian).
- Yu.N.Vigovsky, S.P. Konop, A.N. Malov, S.N. Malov, “Photoinduced phase transitions in layers of dichromated gelatin“, Laser Physics,. 8, N 4, pp. 901 - 915. 1998.
- S.P. Konop, A.G. Konstantinova,A.N. Malov, “Mechanism of the hologram recording in self-developed dichromated gelatin layers”, Photonics & Optoelectron., 3, 21-29, 1995.
- V.P. Sherstyuk, A.N. Malov et al., “Some principles for formation of self-developing dichromate media”, Proc. SP1E, 1238, pp. 218-223, 1989.
- G.N. Zacepina, Water physical property and structure., State University Publ., Moscow, 1998. ( in Russian).
- Method of molecular dynamics in physical chemistry., Nauka, Moscow, 1996. ( in Russian ).
- Yu.N.Vigovsky, A.N. Malov, V.S. Festchenko, “The phase relief formation control in the dichromated gelatin layers”, Computer Optics, No. 17, pp. 75 – 85, 1997. ( in Russian).
- Yu.N. Vigovsky, P.A. Draboturin, A.G. Konop, S.P. Konop, A.N. Malov, “The red gelatin-glycerol recording media with metilen blue dye”, Computer Optics, 18, pp. 133 – 138, 1998. ( in Russian).
|
|
|
|
|
 |
 |
 |
 |
|
Copyright
© 1999-2004 MeDia-security,
webmaster@media-security.ru
|
|
|

